Boyle's law states that the volume of a fixed mass of gas is inversely proportional to its pressure at constant temperature. This relationship means that as volume increases, pressure exerted by gaseous molecules decreases and vice versa
$$ p \propto \frac{1}{v} $$ $$ p = \frac{k}{v} $$ $$ k = PV $$ $$ \text{The mathematical expression is}; $$ $$P_1V_1 = P_2V_2 $$Graphical representation
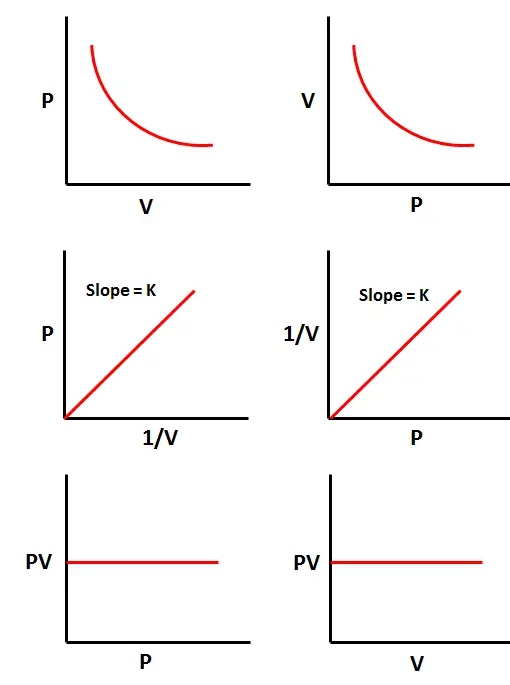
Calculations
Example 1: A gas at a volume \( V_o \) in a container at a pressure \( P_o \) is compressed to one-fifth of its volume. What will be its pressure if it maintains its original temperature, T. (JAMB)
Solution
Example 2: The volume occupied by a gas at 37°C and 740mmHg is 100cm³. What would be the volume of the gas (in cm³) if the pressure is increased to 2000mmHg at the same temperature. (NECO)
Solution
Example 3: A gas occupies a volume of 300cm³ at 410mmHg. What will be its pressure when the volume is decreased to 130cm³ at constant temperature? (NECO)
Solution
Boyle's law Calculator
Formulas:
- For 1: \( P_2 = \frac{P_1V_1}{V_2} \)
- For 2: \( V_2 = \frac{P_1V_1}{P_2} \)