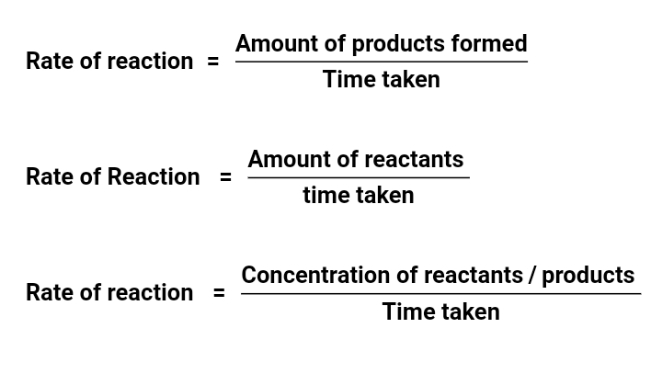
The rate of a chemical reaction is the number of moles of reactants converted or product formed per unit
time. The rate of reaction shows how slow or fast a reaction takes place. For example, the reaction of
sodium and water is fast and spontaneous while the rusting in air is a slow process. A graphical representation of
rate of reaction is called a rate curve.
Collision Theory
The collision theory states that for a chemical reaction to occur, the reacting particles must first collide
with each other. For a reaction to occur there has to be effective collisions between the reacting
particles in order to form products. The conditions for products to be formed are:
- The collisions must be effective
- All molecules should possess a minimum amount of energy to form product molecules
- The right orientation of reactant molecules towards each other.
Activation Energy
Activation energy is the minimum amount of energy that must be possessed by a colliding reacting particle for a
chemical reaction to occur. For a reaction to existing bonds between reacting particles must be broken down
first, this internal bond energy is called an Energy barrier. For a reaction to occur there must
be sufficient energy to break this energy barrier. If the energy is less than the activation energy, no
reaction will occur. Reaction only occurs if the reacting particles possess an energy equal or more
than the activation energy.
Every reaction has its peculiar activation energy. A chemical reaction with a low activation energy occurs
spontaneously. For those with a higher activation energy, they need to be supplied with energy in the form of
heat, light, or electrical energy. A catalyst lowers the activation energy of a reaction.
Endothermic Reaction
An endothermic reaction is a reaction that absorbs heat from its surrounding. The reacting vessel feels
cold to touch. The enthalpy change (heat change) is positive (∆H = -ve) due to the absorption of heat energy
into the system.
.png)
Examples of endothermic reactions include:
- Photosynthesis
- Dissolution of ammonium chloride in water
- Dissolution of salt in water
- Melting of ice to water
- Thermal decomposition
Exothermic Reaction
An exothermic reaction is a reaction that gives off heat to the surrounding. The reacting vessel feels
hot to touch due to the release of heat. The enthalpy change is negative (∆H = -ve).
.jpeg)
Examples of exothermic reactions include:
- Dissolving metal e.g Sodium in water
- Reaction of metals and dilute acids
- Reaction of strong acid with water
- Reaction of acid and a base
- Rusting
- Slaking of lime
- Combustion reactions

.png)
.jpeg)