Based on the collision theory, reactions depend on the frequency of the effective collisions beteen the reacting particles. These factors generally determine the energy content of the reaction thereby controlling the rate of reaction. The factors that affect the rate of a chemical reaction include:
- Nature of reactants: The rate of a chemical reaction is determined by the nature of the particles that react together. For example, the reaction of Zinc with dilute Hydrochloric acid liberates hydrogen gas faster than with a reaction of Iron and dilute Hydrochloric acid which liberates hydrogen slowly. This is due to the reactivity of the metal. Zinc which is more reactive that Iron reacts faster. A piece of Gold will not displace hydrogen from dilute hydrochloric acid because it is lower than hydrogen.
- Concentration: The more concentrated the reacting particles are, the faster the tendency to react to form products. For example, neutralization occurs more rapidly between a concentrated acid and an alkali than with a dilute acid and an alkali.
- Surface area of contact: The greater the contact between the reacting particles, the faster the reaction. In order to achieve a faster reaction, solid particles can be ground into dust or powdered form. Reacting particles should also be thoroughly stirred to increase contact. For example, powdered marble(CaCO3) reacts faster with dilute acids than as marble chips.
- Pressure: This factor only affects gases. THe greater the pressure exerted on the reaction system by gaseous particles, the faster the reaction. For example, a mixture of hydrogen and chlorine gases will react twice as fast if the partial pressure of hydrogen and chlorine is increased from 0.5 to 1atm.
- Temperature: The higher the temperature, the faster the chemical reaction. Since heat is a form of energy, heat is supplied as energy to the reacting particles thereby increasing the active collisions. Reducing the temperature or cooling the reactants will inturn reduce the rate of chemical reaction. This is evident in refrigerators as they prevent spoilage by slowing down the chemical reaction of microbes that causes food spoilage. For example, the reaction of calcium and water to liberate hydrogen gas occurs rapidly with warm water than with cold water.
-
Light: Some reactions are influenced by light. This reactions are called photochemical
reactions. In this reaction, light supplies energy to the reacting particles thereby increasing
their effective collisions.
For example, the reaction of hydrogen and chlorine is explosive in bright sunlight, moderate in daylight and
negligible in the absence of light. Other photochemical reactions include:
- Photosynthesis
- Reaction of chlorine and methane
- Decomposition of hydrogen peroxide
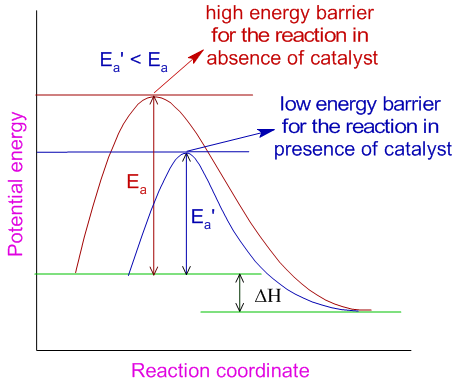
- Catalyst: A catalyst can be defined as a chemical substance that alters the rate of a chemical reaction, but itself remains chemically unchanged at the end of the reaction. Catalysts can speed up or lower the rate of a chemical reactions. Positive catalysts or promoters increase the rate of chemical reaction while negative catalysts or inhibitors reduces the rate of reaction. Generally, catalysts acts to reduce energy barrier by lowering the activation energy of the reaction system.
Properties of catalysts
- A Catalyst alters the rate of chemical reactions
- Catalysts do not determine the type of products formed
- A catalyst remain chemically unchanged at the end of the reaction
- Catalysts are specific in action
- A catalyst cannot start a reaction
- Catalysts do not affect equilibrium position or equilibrium of reversible reactions
Some reactions involving catalysts include:
| Reactions | Catalyst used |
| Hydrogenation of oils to fats
i.e production of margarine |
Finely divided Nickel or Palladium |
| Production of ammonia
Haber process |
Finely divided Iron |
| Esterification | Concentrated sulphuric acid |
| Thermal decomposition of potassium trioxochlorate(V) or hydrogen peroxide | Manganese (IV) oxide |
| Proudction of trioxosulphate (VI) by contact process | Vanadium (V) oxide |